Description
The COVID-19 (SARS-CoV-2) IgG ELISA is intended for the qualitative determination of IgG class antibodies against SARS-CoV-2 in human serum or plasma (citrate, heparin) to support the diagnosis of COVID-19 disease and constitutes a supplement to direct pathogen detection.
Principle of the Covid 19 test
The qualitative immunoenzymatic determination of specific antibodies is based on the ELISA (Enzyme-linked Immunosorbent Assay) technique. Microtiterplates are coated with specific antigens to bind corresponding antibodies of the sample. After washing the wells to remove all unbound sample material a horseradish peroxidase (HRP) labelled conjugate is added. This conjugate binds to the captured antibodies. In a second washing step unbound conjugate is removed. The immune complex formed by the bound conjugate is visualized by adding Tetramethylbenzidine (TMB) substrate which gives a blue reaction product. The intensity of this product is proportional to the amount of specific antibodies in the sample. Sulphuric acid is added to stop the reaction. This produces a yellow endpoint colour. Absorbance at 450/620 nm is read using an ELISA Microtiterplate reader.
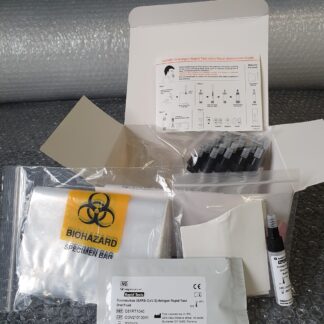
Material included with the covid 19 test
- Microtiterplate: 12 break-apart 8-well snap-off strips coated with SARS-CoV-2 antigens; in resealable aluminium foil.
- IgG Sample Dilution Buffer
- Stop Solution
- Washing Buffer (20x conc.)
- Conjugate
- TMB Substrate Solution
- Positive Control
- Cut-off Control
- Negative Control