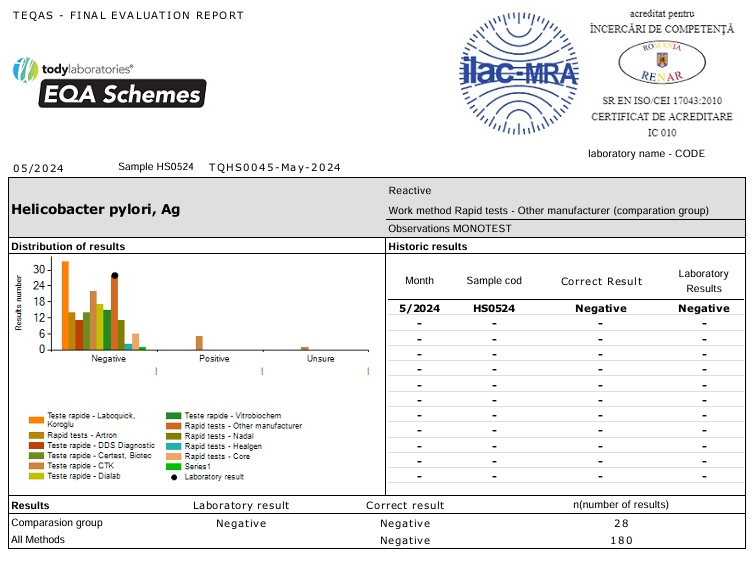
EQA programs are invaluable tools used by laboratories to periodically assess their performance by means of interlaboratory comparisons.
Results are objectively compared between laboratories using:
- the same working method or different methods;
- the same equipment or different equipment;
- the same producer of reagents or different producers.
Comparison of all results provides insight into performance that is not available to the individual participants. Moreover, the information offers a basis for quality improvement initiatives.
EQA evaluation reports also offer a comparison of different methods and equipment for the same test; thus, providing an assessment of reliability. Besides the use of daily quality controls, the external quality programs enhance confidence in reporting patient test results
For this purpose we collaborate with the company Todylaboratories which provides the samples.
Advantages
- EQA Schemes are fully accredited to help meet the current regulatory needs of
clinical laboratories. - the evaluation reports with Z score calculation and graphical representation of result’s Bias to facilitate monitoring the evolution over time of this essential perfor mance indicator;
- ranges for the following areas: Biochemistry, ESR, Gynaecological Pathology Hematology, HIV Antibodies, Hepatitis A, Hepatitis B+C, Hormones and tumor markers, Infection Disease Serology, Mycology, Microbiology, Parasitology, Preanalytical phase, Transfusion – Immunohematology, Urine Chemistry.